The High-Impact Medical Journal Editors Harassment Of The World's Leading Clinical Researcher of Repurposed Drugs in the COVID Pandemic - Part 1
The FLCCC's Flavio Cadegiani performed one of the highest-quality studies in COVID, finding unprecedented reductions in mortality with proxalutamide. He has been under attack ever since.
If you’ve read my first 3 posts on the corrupt actions of the high-impact journal ‘Editorial Mafia’ (here, here, and here), buckle up, because the attacks on researchers of repurposed drugs gets way worse. None more so than the numerous academic, political, psychological, and legal actions taken against Dr. Flavio Cadegiani. The next four posts will detail both the fragmented and co-ordinated attacks on Flavio and his team as they researched numerous therapeutics during COVID, but none triggered more harassment than their work on the drug proxalutamide. This series will culminate in the detailing of severely threatening actions taken against him in Part 4.
Although I will focus on the harassment of Flavio Cadegiani, I do want to mention the impressive team of researchers he worked with on their studies of anti-androgens as they too have suffered harassment. The co-authors are listed in his two proxalutamide publications here and here.
I focus mostly on Flavio as he was both the lead author and had the most prominent public profile, thus the attacks have largely targeted at him. The corruption of the high-impact journal actions against proxalutamide not only exceed those of any of my prior posts on their actions against ivermectin, but after the proxalutamide papers were finally successfully published in a lower-tier journal, the attacks went beyond the journals into political, personal, and physical harassment. Let’s go.
One difference with Dr. Flavio Cadegiani’s four (yes, four) high-impact journal rejections/retractions is that the studies did not involve ivermectin. Instead, his studies involved a novel prostate cancer medicine called proxalutamide that inhibits testosterone, a mechanism that was highly effective in the prior variants when SARS-CoV2 relied on an enzyme (TMPRSS2) stimulated by testosterone to enter the cell.
Flavio and his colleagues figured out that if you blocked any hormone with testosterone activity, you blocked SARS-CoV2’s entry and replication. He first tested this hypothesis in a series of observational studies, where he consistently found a correlation between “androgen blocking” (testosterone and its metabolites) medicines and improved outcomes. Conversely, in these studies, the team also found that androgen excess led to worse outcomes. They were onto something and they knew it. They had identified an important pathophysiologic mechanism which was then further supported by robust observational studies indicating that hormonal blocking would act as a potent therapeutic in both men and women ill with COVID-19.
Flavio and team did everything they could to prove, in a rigorous (and I mean rigorous), prospective manner, the efficacy of a drug with strong anti-androgen activity. The reason they chose to study proxalutamide was that it was the only medicine they could obtain without spending their entire life savings and that also had a physically identical placebo tablet (this is really important when conducting blinded clinical trials). Just ask Ed Mills and the rest of the corrupt NEJM TOGETHER ivermectin trial investigators or the corrupt JAMA Lopez-Medina trial investigators if their placebo tablets looked like their active study drug tablets. The idea that these guys are supposedly respected trialists is absurd.
Curiously, the only drug they could study came from a non-Western Pharma company called Kintor. All of the western Big Pharma companies (Jannsen, Pfizer and others) refused to support his study and Flavio told me that at one point they tried to discourage him from studying anti-androgens at all.
As any reader of mine knows by now, the modern rules of medical science state that to “prove” something works, you need a “Big RCT” (randomized controlled trial). Although this “requirement” is based on a scientific myth, it is unfortunately a political truth. The Pharma-conflicted masters of “evidence-based medicine” have inculcated this notion into what is now three generations of physicians. But since them’s the rules, Flavio and his colleagues knew they had to play by them. Their observational studies, as convincing or numerous as they may have been, were not going to move the needle. At all.
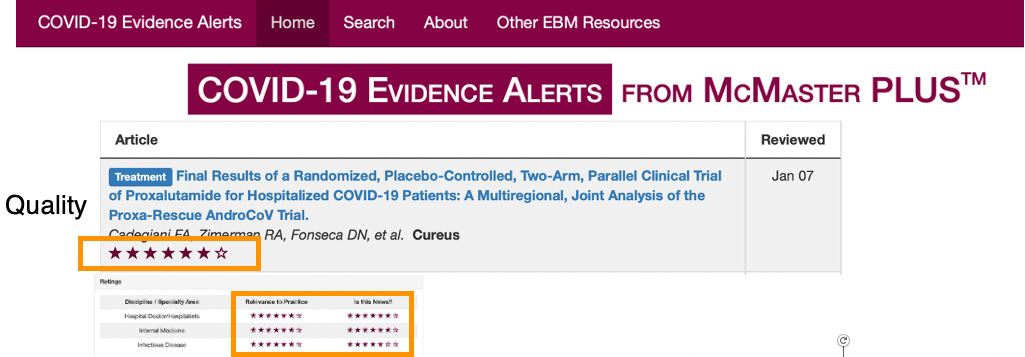
So they set up conducting the highest quality trial in the pandemic (based on the most established grading scale of clinical trials), as it was a prospective, multi-center, double-blind, randomized controlled trial (PMCDBRCT). The rating of this trial was done by COVID-NMA, an international initiative led by a number of supposedly august institutions:
Here is their grading, taken from a slide talk that Flavio gave:
Even Ed Mill’s University (McMaster) gave it a superb quality rating.
Their study was supposed to have been an evidence-based maniac’s wet dream. Especially since it found that treatment with proxalutamide led to massive improvements in survival, hospital length of stay, and recovery rate. Proxalutamide-treated patients had an 8.0% mortality vs. 39.2% in the placebo group. The same benefits were found in men and women.
This was an almost unheard-of improvement in survival difference amongst the medicines and diseases that I have studied. The “number need to treat” (NNT) to save one life comes out to 3.2 patients. This rivals the potency of defibrillating someone who is in the midst of a cardiac arrest (NNT = 2.5 - a lower number reflects higher potency).
I say the NNT is almost unprecedented because the difference in mortality that they found is identical to the mortality difference found by Dr. Paul Marik in his 2017 observational trial of IV Vitamin C in severe sepsis patients (8.5% vs 40.4%). Not so fun fact: Professor Marik’s study is currently in the process of being retracted by Paul’s ex-hospital as the result of complaints from people who have been attacking ivermectin. This retraction is underway, now 5 years after publication and despite the fact his co-authors still work for the hospital. As below, “they” will do everything they can to ruin our credibility and career accomplishments.
In comparison to Paul’s study, Flavio’s trial was a larger, prospective, randomized trial which included 778 patients. It was conducted in Brazil during one of the most violently virulent variants (a mouthful I know) in the pandemic. Fortunately, the Gamma variant “burned itself out” in South America without spreading globally to an appreciable extent given that Gamma was up to four times more lethal than Delta. Whoa. If it had spread internationally to a significant extent, it would have caused a global apocalypse given that most of the world still relies on an absence of early treatment, an anemic dose of corticosteroids, and an absurd reliance on hospital-phase infusions of Run-Death-Is-Near. The lethality of the variant explains the high mortality rate of 3,500 to 4,000 deaths per day in Brazil during the study time period.
Three factors turned the Gamma variant into a national catastrophe for Brazil but not the world. Gamma had lower transmissibility and Brazil’s borders were closed at the time. But, they were forced to transfer severely ill patients nationally out of Amazonas after Gamma emerged there due to the fact that the Amazonas health system had been over-run and was in a state of collapse.
Prior to designing and conducting his study, Flavio negotiated with the manufacturer that if the drug proved beneficial, the company would agree to sell the drug for just $11 a dose. Instead of receiving money from the company to conduct the trial, he understood that during a global health emergency crisis, saving lives mattered more than making money and he agreed to conduct the trial for free. Let that sink in for a second. He paid, from his own pocket, all the operating and hospital costs of the trial using only minor donations from local businessmen. Kintor did donate the supplies of the medicine, but again, the arrangement was contingent on the fact Kintor was obligated to keep it affordable on a worldwide level.
Notice this approach is the opposite of what typically happens in medical research. Normally Pharma pays off investigators to manipulate trials into showing their drug works and then they charge exorbitant prices to governments and health systems around the world.
Another important action that Flavio and his study team must be commended for is that they immediately made public the de-identified individual patient level (IPL) data of all the subjects in their study (they do this with all of their studies). This action is what experts have mandated for decades but absolutely no pharmaceutical company ever does. Ever. Remember the TOGETHER ivermectin trial? Those investigators wrote over and over in their protocols and publications that their IPL data would be made public immediately upon completion of the study. Not-so-fun fact: they immediately backtracked after publication in the NEJM and now refuse to make their data public despite repeated requests by researchers from all over the world.
If you knew Flavio, you would know his entire mission and focus during COVID was in figuring out which drugs worked (and he found, studied, and “proved” the efficacy of a number of them). Just look him up on Pubmed. My guess is that he is the most diversely published clinical researcher on therapeutics in COVID in the world. But he didn’t care about any single drug. Although he is a world expert on ivermectin in COVID and knows it works, he also knows a lot of other compounds that work too. This is why his original protocols in Brazil, like those of the FLCCC, the American Association of Physicians and Surgeons, and Truth For Health, have always employed combination therapy protocols. It’s not just about ivermectin and it never was.
What follows is a highly detailed compilation of the numerous corrupt actions by four different journals — including three of the highest-impact journals in the world. Numerous emails will show the brazen and unprecedented actions taken to both delay and avoid publishing the results of his trial. Ultimately, the cumulative actions delayed publication for 18 months until it was finally published in a lower-tier journal that has been largely (but not completely) ignored. Mission accomplished.
First Submission: New England Journal of Medicine (NEJM), April to June, 2021
Flavio first submitted his paper to the highest-impact medical journal in the world in April of 2021. Again, his was a high-quality study which found a massive reduction in mortality. Recall that in the original study of Remdesivir, the investigators reported a brief (a couple of days) reduction in hospital length of stay and no impact on mortality. Yet Fauci immediately called an Oval Office meeting, invited legions of the press, and extolled the drug as a “game-change-uh.” This performance was immediately broadcast across the world via the internet, radio, newspapers, and televisions. It even made the front page of the New York Times — all before it was peer-reviewed or published. Fauci effectively launched a global PR campaign for a pharmaceutical company… for free. The US then spent over $3 billion to purchase Remdesivir in its first year. On an anti-viral relegated for use in a phase of the illness where live virus is rarely present. The good ole’ United States of Pharma.
With proxalutamide, let’s say that things went a little differently (understatement of the century). Given the trial’s size, quality, and magnitude of its findings, the NEJM had to send it out for peer review. Well, let’s not say they “had to” but they did. The peer reviewers began submitting their questions and suggested revisions to Flavio and his co-authors. The team answered all the reviewers’ questions and incorporated the suggested revisions. The reviewers had no more questions. So, accept and publish right? Oh, that’s so 2019. Two months after submission, Eric Rubin, the journal’s Editor-in-Chief, wrote this reply:
Remember Dr. Rubin? He was the one who told a NYT Magazine writer that I “got lucky” when I testified in Senator Ron Johnson’s first Senate hearings on COVID in May 2020 about how corticosteroids were critical in the treatment of hospitalized COVID patients. Two months later, corticosteroids became the standard of care worldwide. Lucky Pierre.
Now, not-so-fun fact about Eric Rubin and the NEJM. When Flavio asked if reviewing patient-level data was a routine part of the process for being published in the NEJM, Dr. Rubin admitted that the NEJM had never analyzed the primary raw data of any study before publication in its history. Interesting, no? The standard practice of the high-impact journals responsible for publishing the now-proven fraudulent vaccine trials, as well as those of Paxlovid and Molnupiravir, was to do so without analyzing the patient-level data on which the manuscripts’ conclusions were based. Yet here, suddenly, they “made up a new rule” to reject Flavio’s study of an $11 medicine.
So, the moral of the story is that the journals trust Pharma… but don’t trust independent investigators without conflicts of interest?
“Sorry, we are not interested in publishing your study showing a massive reduction in mortality.”
Flavio wrote back:
Rubin responded:
“Too good to be true?” That’s the excuse? Meanwhile, Pfizer can claim an 88% reduction in hospitalization in high-risk COVID patients treated with Paxlovid, but that doesn’t need verifying. Isn’t that curious, given the fact that Paxlovid failed to show efficacy in low-risk patients as well as in prevention but somehow it led to an 88% reduction in high-risk patients? I would think that should have been checked out, no?
Let’s compare the discordant results among Paxlovid trials with those of proxalutamide. In their other study testing proxalutamide in mild to moderately ill Covid outpatients, the time to clinical recovery was 1.8 days with proxalutamide vs. 12 days with placebo. Whoa. Also, by day seven, 82% of proxalutamide-treated patients were PCR negative, while only 31% of placebo were. Not. Even. Close.
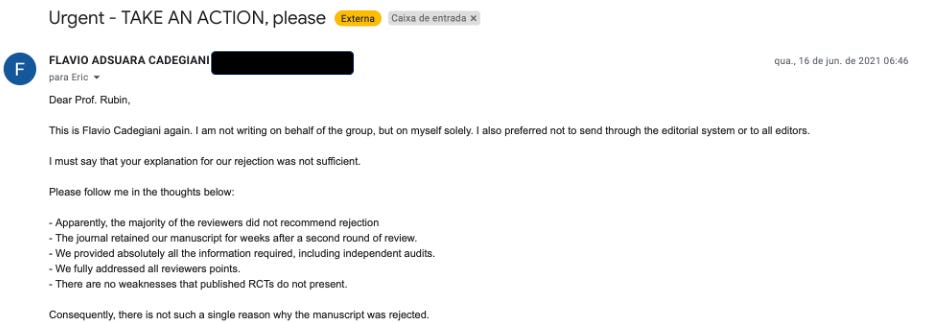
Flavio decided to make a desperate plea to Rubin, pointing out the most frightening aspect of his and the journal’s action, which is that it would lead to many people dying as a result.
So, Flavio’s paper passed standard peer review but was instead effectively retracted by the NEJM editor Eric Rubin. Using one of the lamest, made-up reasons to do so. Absurd and laughable, except for all the people who subsequently died because of the suppression of the efficacy of a medication whose “trial results were too good to be true.”
Recall that the PFDA (the P is not a typo) went to court to hide Pfizer’s vaccine IPL data for 75 years. The judge wasn’t buying it, and mandated the release of the IPL data. Yet, the NEJM had not reviewed Pfizer’s IPL data before publishing. I can also guarantee that the PFDA Committee members issuing the EUA did not review Pfizers IPL data to any significant extent because the EUA was a foregone conclusion, just like their unanimous vote to jab toddlers with COVID mRNA injections (I literally get nauseous every time I think of that vote and the data it was based on). That is why they are called the PFDA. We now know from Ventavia whistleblower Brook Jackson’s reports as well as the work of Naomi Wolf and the thousands of volunteers at the Daily Clout, that the study was rife with fraudulent manipulations and obscuring of adverse data.
So not only did Flavio share his IPL data, but shared it after conducting one of the highest-graded quality trials since the pandemic began (although I champion his achievement, in reality, quality grades have no correlation with the accuracy of a study’s conclusion).
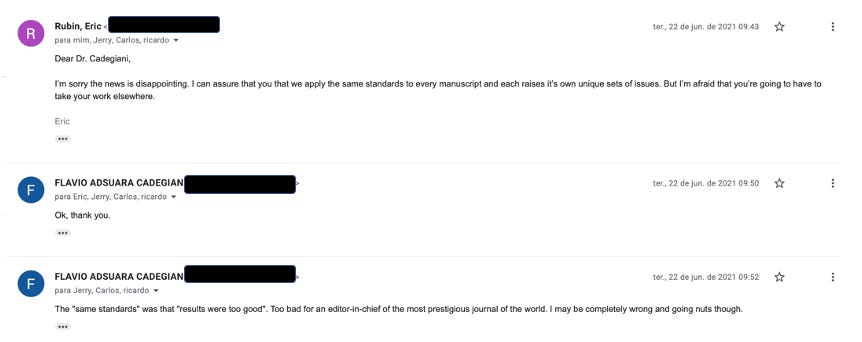
Rubin’s reply to Flavio’s final plea on behalf of humanity?
“I am afraid you are going to have to take your work elsewhere.”
Flavio was NOT going nuts. At all.
Second Submission: The Lancet - June 11, 2021 to June 22, 2021
Flavio et al then submitted to the second highest-impact journal in the world. Again the paper was sent out for peer-review (which is the biggest hurdle in publishing manuscripts, and to achieve this feat at the two top journals in the world is beyond impressive, which speaks to the quality and size of his trial).
Flavio and his team received the reviewer comments very quickly, and before they had an opportunity to respond to them (as is customary) it was rejected at the editorial level. This despite the fact the issues and questions raised by the reviewers could have been easily addressed and further, no peer-reviewer had recommended rejection in their comments (I read their comments). Curious no? The Editors’s reason was that they wanted to give “priority to other manuscripts under consideration.” I am sure those manuscripts also showed massive impacts on mortality in a novel disease engulfing the globe. My God.
Manuscript reference number: THELANCET-D-21-04718
Title: Efficacy of Proxalutamide (GT0918) in Hospitalized COVID-19 PatientsDear Dr. McCoy,
Thank you for submitting Efficacy of Proxalutamide (GT0918) in Hospitalized COVID-19 Patients to The Lancet. This is an interesting area of research, so we sought external views to add to our own.
The reviewers' comments were mixed. After discussing the paper further, we were unable make this submission a priority for our general readers when compared with other papers also under consideration. As a result, I regret to inform you that we have decided to decline this submission.
I am sorry to disappoint you on this occasion and hope that the reviewers' comments, appended below, will help to guide a successful submission elsewhere.
Yours sincerely,
The Lancet
Recall that The Lancet is the same journal that published the fraudulent Surgisphere study of hydroxychloroquine with clearly fabricated data - a scandal that the world has conveniently forgotten about. Good times.
So, Flavio’s study manuscript was first submitted to the two highest-impact medical journals in the world which was rejected arbitrarily by editors despite the peer-reviewers lack of such a recommendation. The journey of this paper to publication is not over. It simply gets increasingly disturbing. Click here for Part 2.
I just want to say thanks to all my subscribers, especially the paid ones! Your support is greatly appreciated as it allows me to devote what is often large amounts of time I spend researching and writing my posts, so again, thanks.
P.S. I opened a tele-health clinic providing care not only in the prevention and treatment of acute COVID, but with a specialized focus on the study and treatment of both Long-Haul and Post-Vaccination injury syndromes. If anyone needs our help, feel free to visit our website at www.drpierrekory.com.
P.P.S We are organizing the world’s first conference on understanding and treating Spike protein induced disease (i.e long haul COVID and vaccine injury syndromes). Tell your doctor to come. Link below:
P.P.P.S. I am writing a book about what I have personally witnessed and learned during Pharma’s historic Disinformation war on ivermectin. Pre-order here for:













i don’t know what the punishment/remedy is for the coordinated destruction of Flavio’s work & reputation by corrupt mortals playing god; however, it should begin with him taking what you wrote to a legal team that includes defamation, fraud & antitrust experts. There’s a firm that comes to mind, and an old friend who at least might listen to Flavio.
This must be exposed & stopped. You know where to reach me--when you return from Orlando.
These evil people are playing a dangerous game.
“game-change-uh”
I suppose having the ability to cloak mass murder under CDC protocols was indeed a game-change-uh! 🤣
Thank you for informing us about proxalutamide—I had never even heard of it! And it doesn’t appear to be on John Droz’s radar (https://c19science.info/FDA_Drug_Approvals2.htm) either—maybe because the research has been thwarted/suppressed, so there isn’t enough to merit inclusion on his list?
I loved hearing about your own journey toward becoming a doctor, Pierre. I wonder if the fact that you came to this career later in life has made you less vulnerable to the groupthink that most doctors succumb to (besides your unassailable integrity and innate sensitivity to propaganda).